贵州大学池永贵/吕文心Nat. Commun.:利用三氟甲基铜(III)配合物作为二氟卡宾前体实现糖类和复杂醇类的二氟甲基化反应
2026-04-14 浏览次数: 10
贵州大学池永贵/吕文心Nat. Commun.:利用三氟甲基铜(III)配合物
作为二氟卡宾前体实现糖类和复杂醇类的二氟甲基化反应
摘要:Grushin’s reagent, (bpy)Cu(CF3)3, is a well-known trifluoromethylation agent, but its potential as a difluorocarbene source has remained largely unexplored. Here, we present a photo- and acid-mediated strategy that repurposes Grushin’s reagent as an efficient difluorocarbene precursor for the difluoromethylation of diverse alcohols, including complex primary, secondary, and tertiary alcohols bearing multiple polar functional groups. This method exhibits broad functional group compatibility and has been successfully applied to the late-stage modification of complex natural products and bioactive molecules. A notable achievement is the regioselective difluoromethylation of saccharides and polyols, a challenging transformation enabled by Me2SnCl2, which serves a dual role as a hydroxyl activator and an in situ source of hydrogen chloride. Antifungal activity evaluation reveals that compounds 3c and 3n possess good efficacy, highlighting their potential as promising leads for antifungal development.
研究背景:图4. 糖类和多元醇的二氟甲基化反应底物范围 (图片来源于Nat. Commun.)
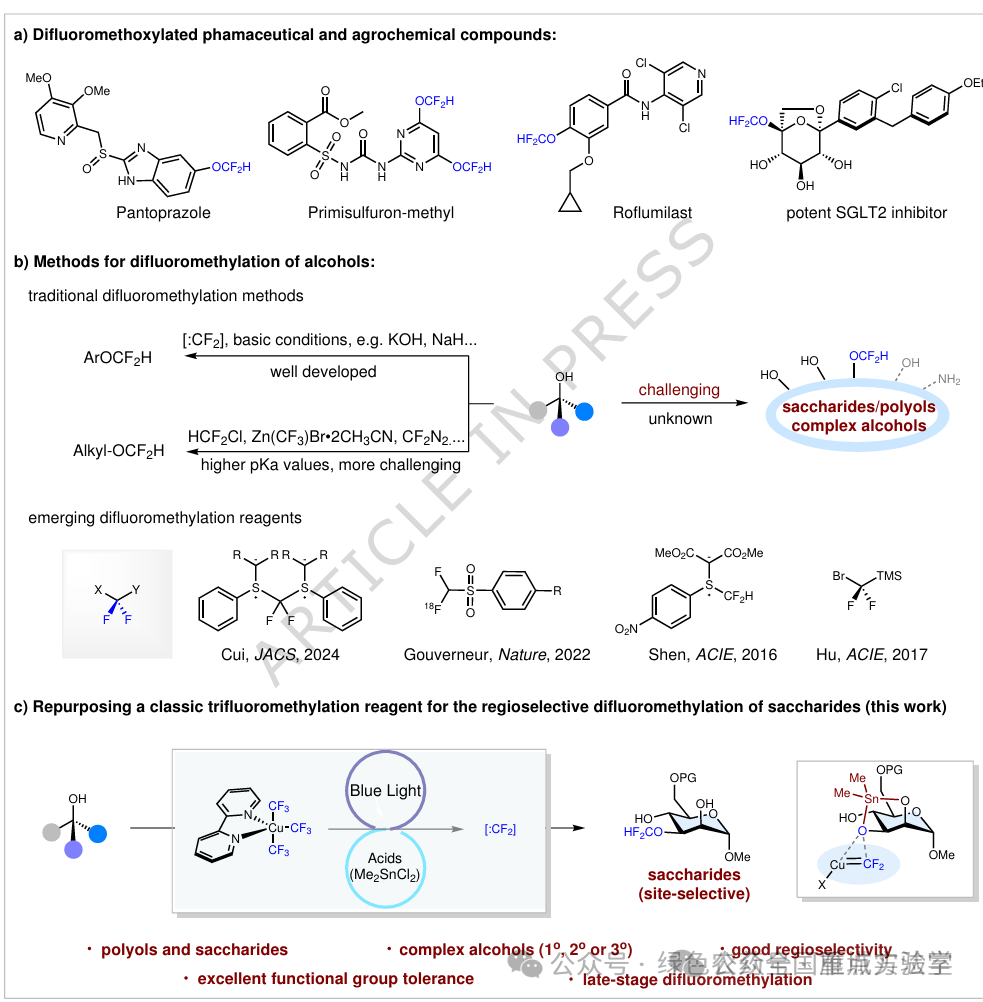

图1.研究背景及策略(图片来源于Nat.Commun.)
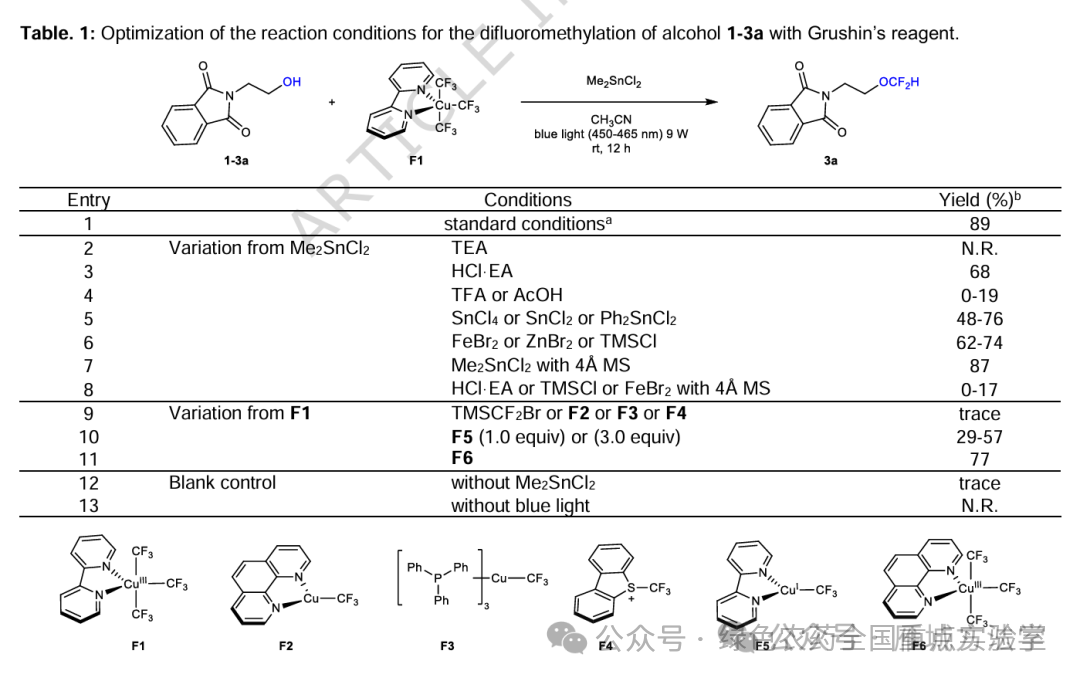
图2. Grushin’s试剂实现醇1-3a的二氟甲基化反应条件优化
(图片来源于Nat. Commun.)

图3. Grushin’s试剂实现糖类1-2j的区域选择性二氟甲基化反应条件优化
(图片来源于Nat. Commun.)
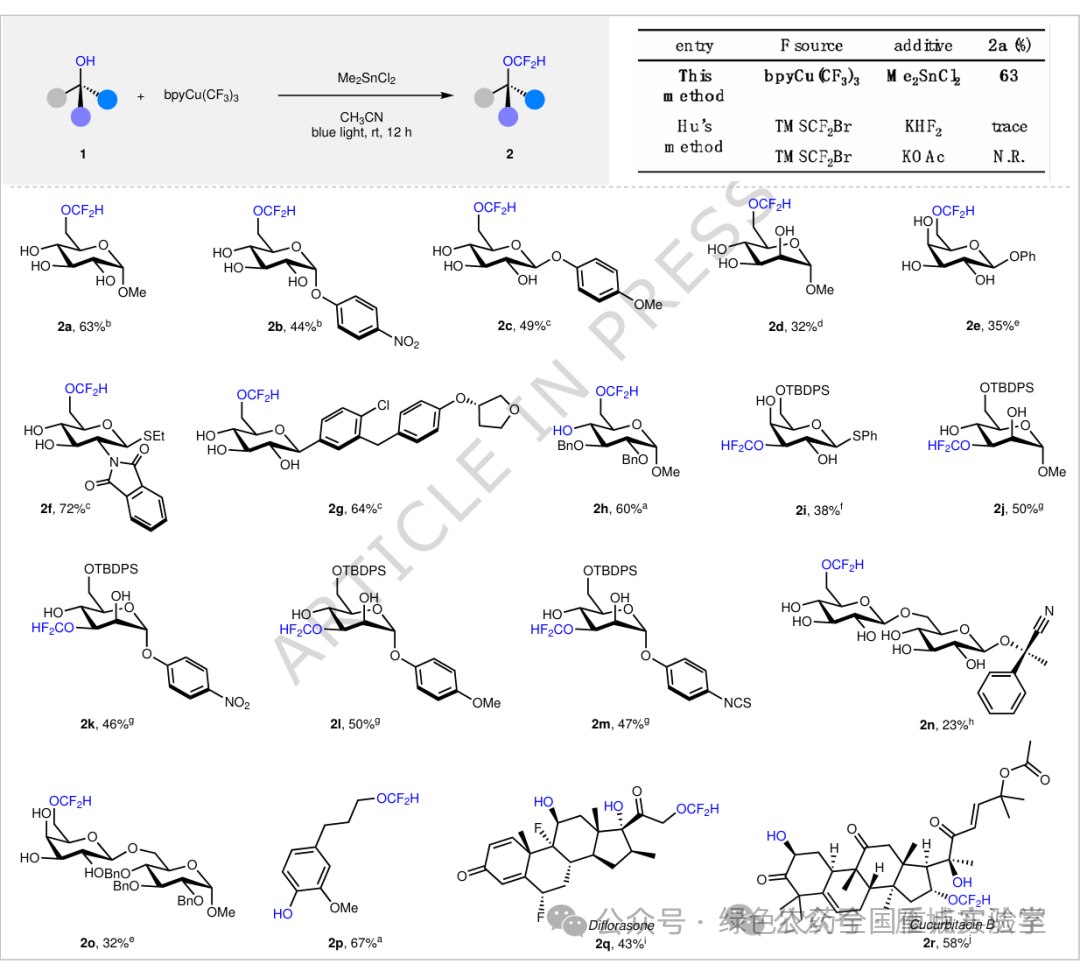

图4. 糖类和多元醇的二氟甲基化反应底物范围 (图片来源于Nat. Commun.)
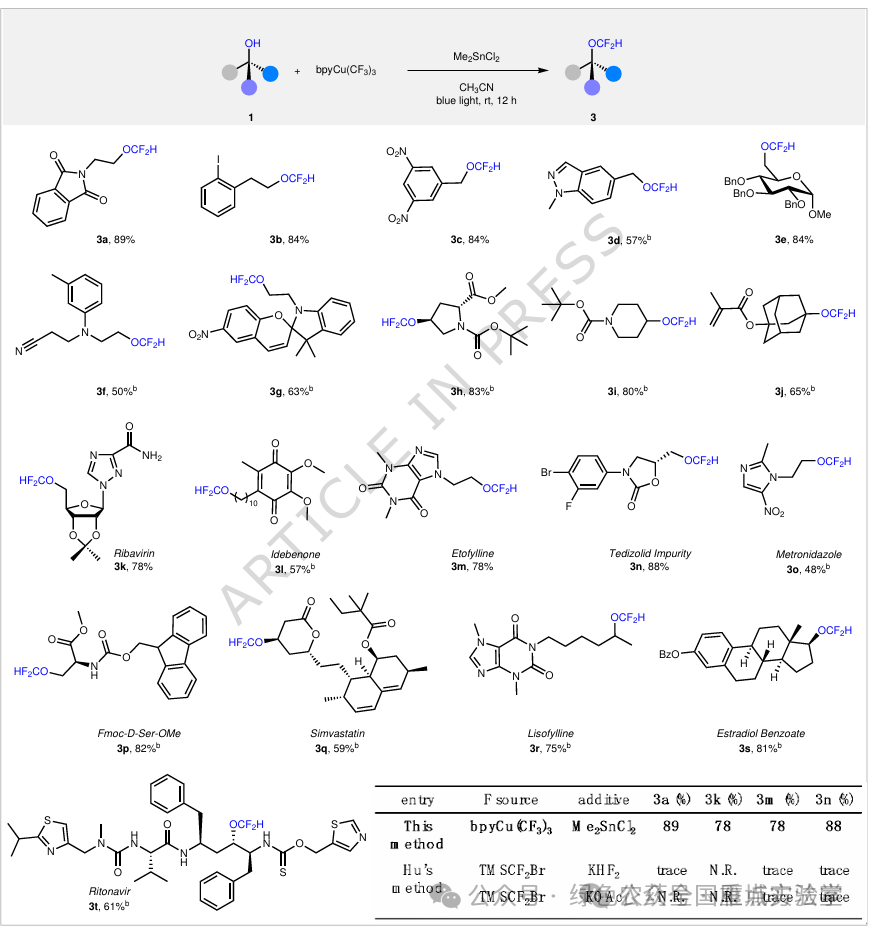

图5. 伯、仲、叔醇的二氟甲基化反应底物范围 (图片来源于Nat. Commun.)
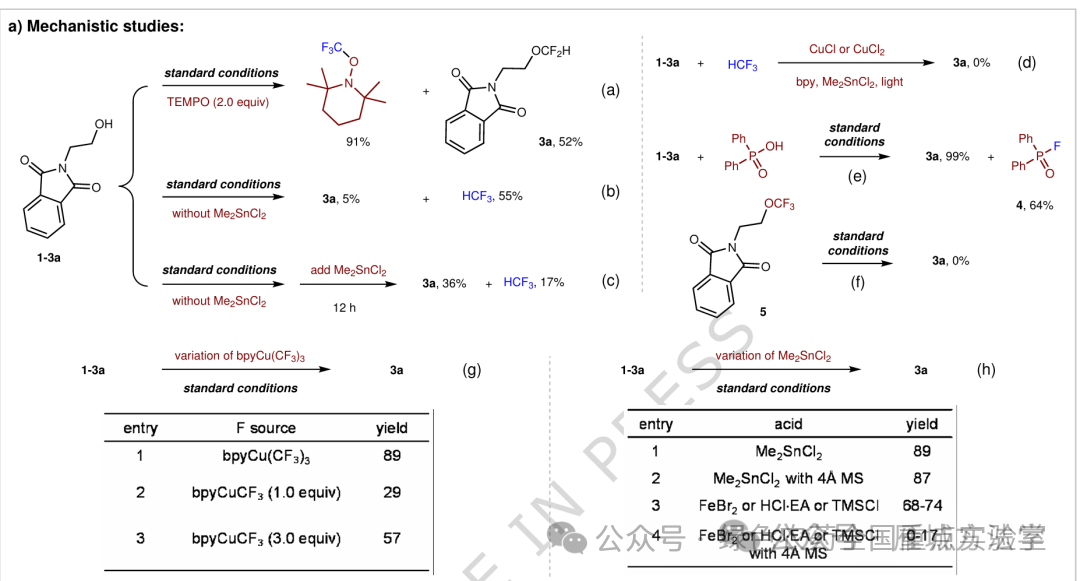
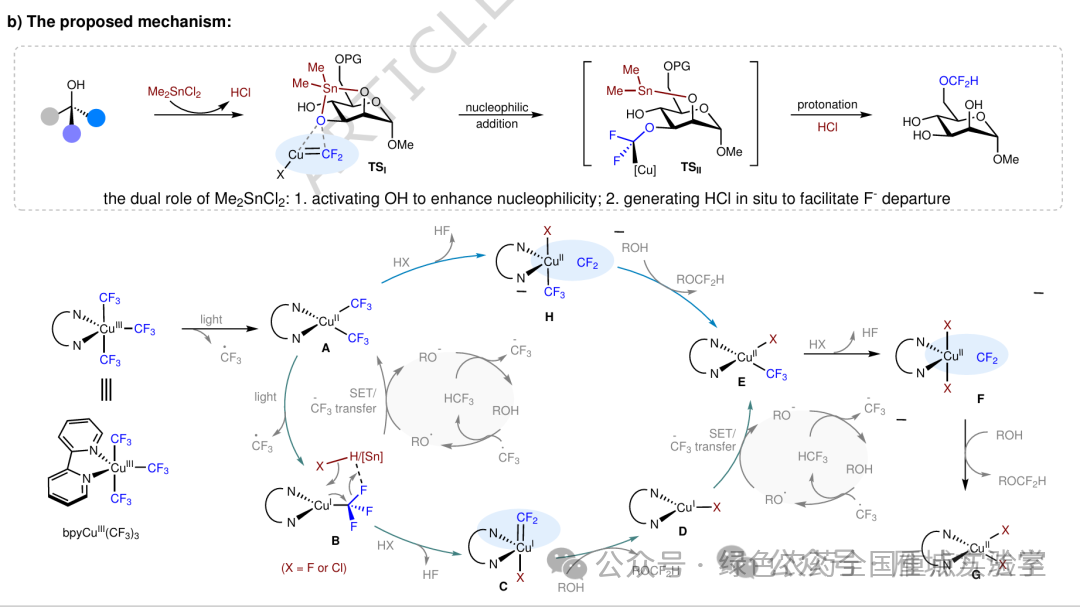
图6. 机理研究及可能的催化循环 (图片来源于Nat. Commun.)
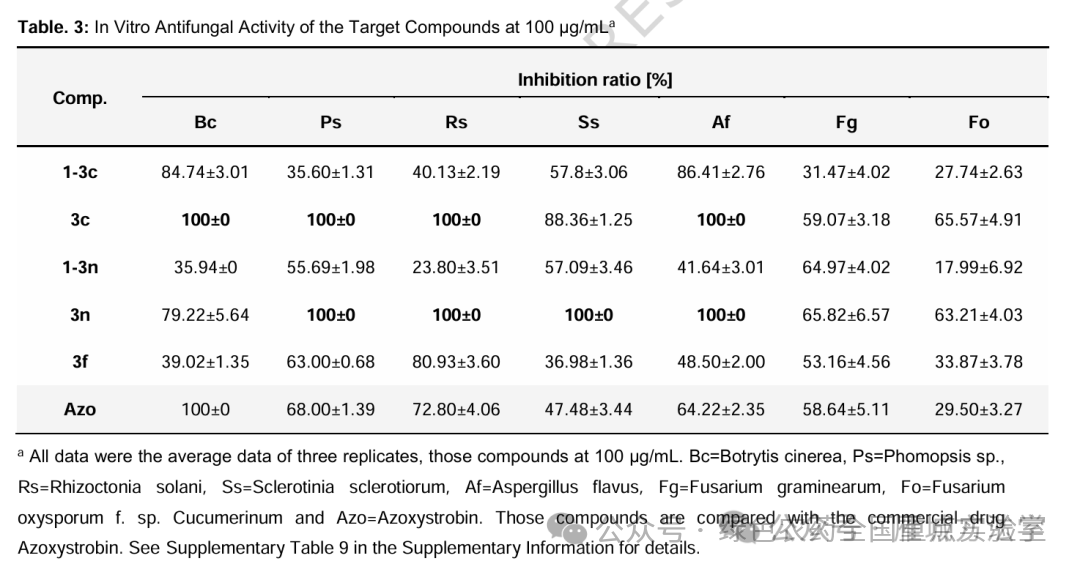
图7. 目标化合物的抗真菌活性测试 (图片来源于Nat. Commun.)
总结:
In summary, we have repurposed Grushin’s reagent as a versatile and efficient difluorocarbene precursor for the photo- and acid-mediated difluoromethylation of saccharides and complex alcohols, including primary, secondary, and tertiary substrates bearing multiple strong polar functional groups. By utilizing Me2SnCl2, which serves dually as an OH activator and an HCl source, this strategy achieves site-selective difluoromethylation of challenging saccharides and polyols, alongside the late-stage modification of complex molecules with broad functional group tolerance. Promising antifungal activity observed in several novel difluoromethyl ether derivatives provides valuable insights for the development of antifungal agents, and research is currently underway to identify candidate molecules with enhanced efficacy.
(转载自:雁城方法学,2026-04-14)
文献信息:

审校:吴增雪
编审:李向阳
终审:张 林