贵州大学伍星星CCL:NHC催化糖基溴代物和醛的交叉偶联反应高立体选择性合成C-酰基糖苷
2026-04-14 浏览次数: 10
贵州大学伍星星CCL:NHC催化糖基溴代物和醛的交叉偶联反应高立体选择性合成C-酰基糖苷


图1. TOC (图片来源于Chin. Chem. Lett.)
摘要:
Replacing hydrolytically labile C–O glycosidic bonds with robust C–C linkages has emerged as a powerful strategy to access structurally stable C-glycosides that mimic native sugar scaffolds. While transition-metal-catalyzed C-acyl glycosylation has significantly expanded synthetic access to bioactive C-glycosides, concise organocatalytic methods that directly convert common glycosyl precursors into stereodefined glycosyl ketones remain scarce. Here we report an N-heterocyclic carbene–catalyzed cross-coupling of glycosyl bromides with aldehydes, providing an efficient route to C-acyl glycosides under mild conditions. The use of simple aldehydes as the acyl source significantly improves step economy relative to protocols requiring preformed anhydrides or thioesters. Mechanistic studies support a radical pathway in which single-electron activation of the glycosyl bromide generates a glycosyl radical that couples with the NHC ketyl intermediate to forge the C–C bond. This strategy delivers products with exceptional anomeric α-selectivity, establishing a mechanistically distinct and highly stereocontrolled organocatalytic platform for the streamlined construction of structurally complex C-acyl glycosides.
研究背景:
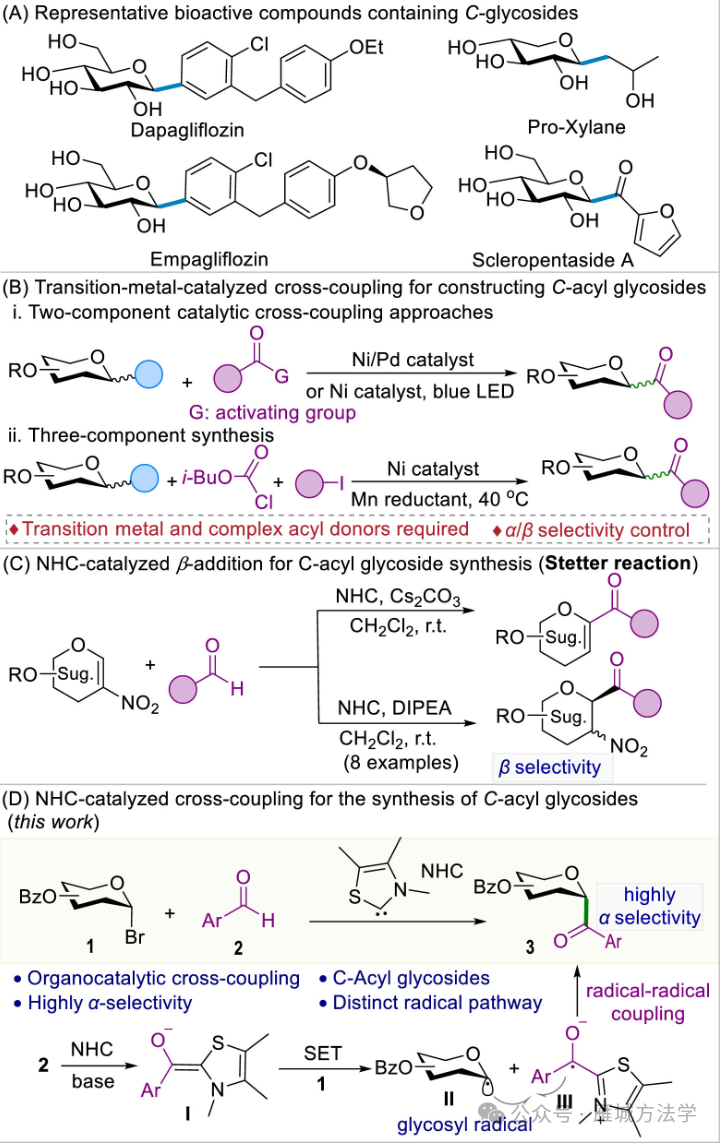

图2. 研究背景及本策略 (图片来源于Chin. Chem. Lett.)
研究内容:
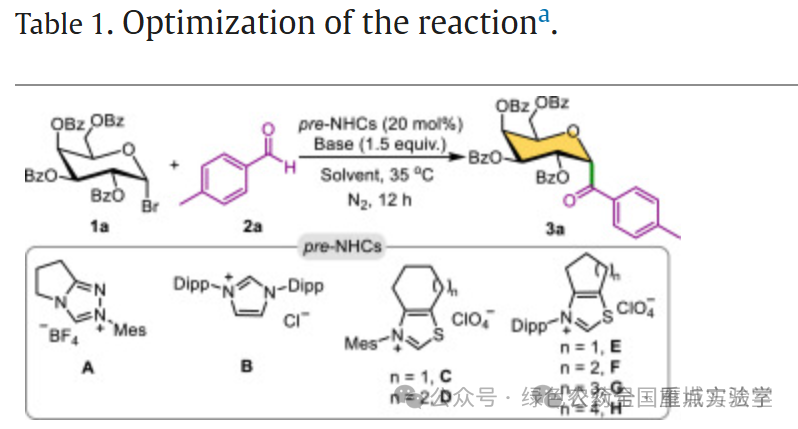
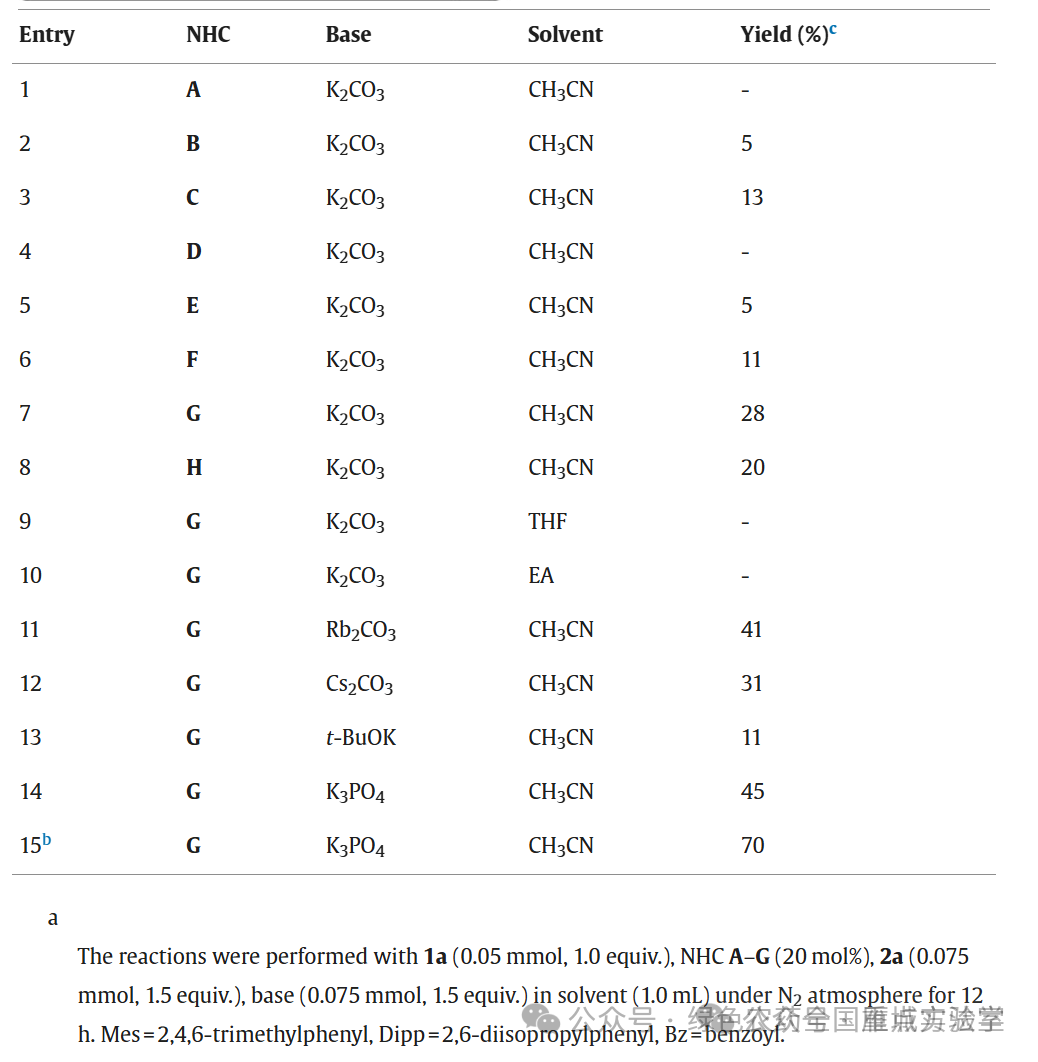
图3. 条件优化 (图片来源于Chin. Chem. Lett.)
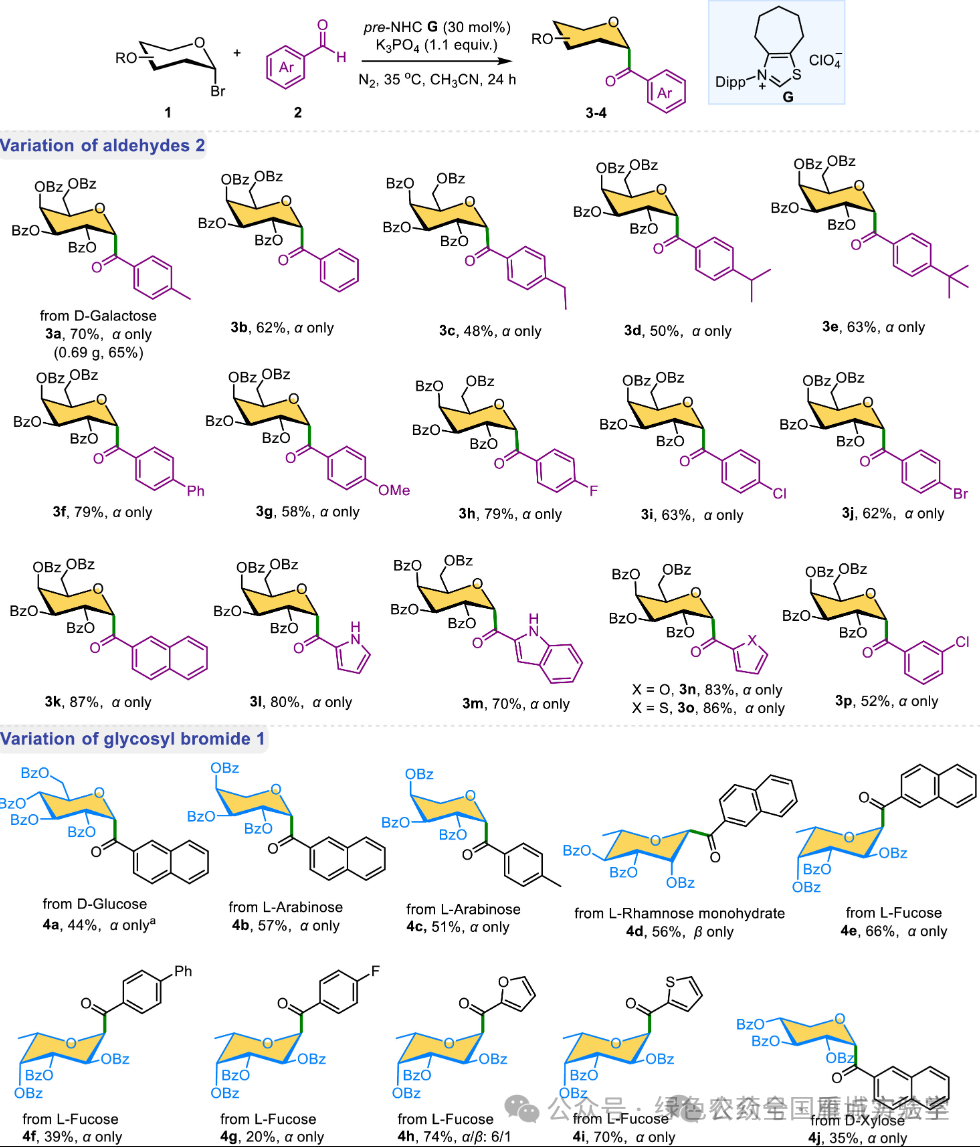
图4. 底物拓展 (图片来源于Chin. Chem. Lett.)
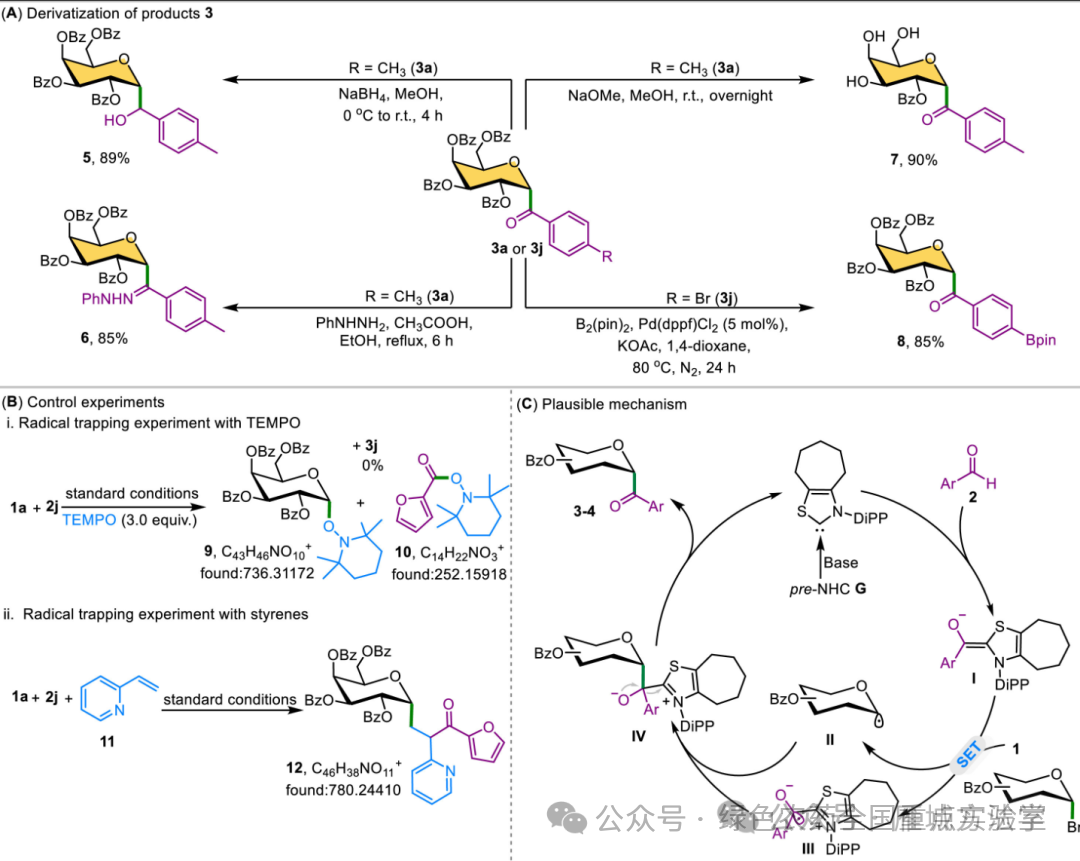

图5. 合成应用、机理研究及可能的催化循环 (图片来源于Chin. Chem. Lett.)
总结:
In summary, we have developed an N-heterocyclic carbene organocatalytic cross-coupling strategy that enables the direct construction of C-acyl glycosides through the straightforward assembly of readily available glycosyl bromides with simple aldehydes. This organocatalytic strategy provides a concise alternative to transition-metal-mediated C-acyl glycosylation and avoid the use of preactivated acyl donors such as anhydrides or thioesters, offering broad substrate generality and excellent diastereocontrol. Mechanistic studies support a radical manifold in which NHC-promoted single-electron transfer from glycosyl bromides generates glycosyl radicals that subsequently engage aldehyde-derived ketyl radical intermediates to forge the C–C bond at the anomeric center, delivering the C-acyl glycosides as single stereoisomers.The C-acyl glycosides accessed herein serve as versatile synthetic linchpins for further diversification toward structurally complex and biologically relevant C-glycoside architectures. Ongoing efforts in our laboratory focus on expanding NHC-enabled radical processes for carbohydrate functionalization and exploring the application of these robust C-glycoside scaffolds in agrochemical discovery.
(转载自:雁城方法学,2026-04-13)
文献信息:

END
更多精彩
关注我们
编辑:邓泽蕊
审校:吴增雪
编审:李向阳
终审:张 林